Management system guidance
6.0 Planning
ISO Navigator Pro™ is a free tool that provides practical, expert guidance for businesses wishing to interpret and better implement the requirements of ISO 9001:2015, ISO 14001:2015 and ISO 45001:2018.
Our range of templates cover the requirements of ISO 9001:2015, ISO 14001:2015 and ISO 45001:2018, and offer an easy way to implement your next management system.
6.3 Planning for change
This is a new requirement for ensuring that once your processes are determined, and their sequence is documented within your quality manual; your organization should identify the risks and opportunities associated with these processes.
In order to realize the benefits associated with the identified risks and opportunities, subsequent changes to the management system may be needed. These changes could relate to any aspect of any process, such as its; inputs, resources, suppliers, personnel, activities, controls, measurements, and outputs.
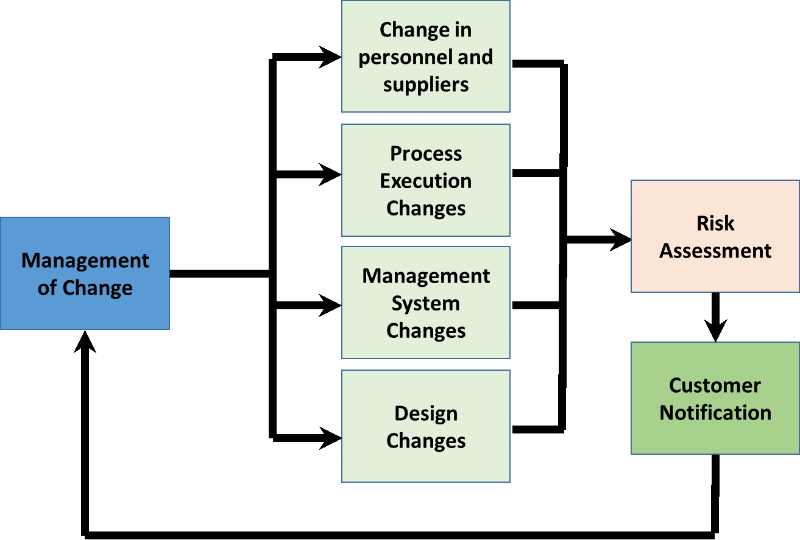
Changes are intended to be beneficial but they need to be carried out when determined by your organization as relevant and achievable. In addition, consideration of newly introduced risks and opportunities should also be taken into account.
To achieve the benefits associated with changes, your organization should consider all types of change that may occur. These changes may be generated, for example, in:
- Processes and procedures;
- Quality manual;
- Documented information;
- Infrastructure;
- Tooling;
- Process equipment;
- Employee training;
- Supplier evaluation;
- Stakeholder management;
- Interested party requirements.
Whenever quality management system changes are planned, Top management should ensure that all personnel are made aware of any changes which affect their process, and that subsequent monitoring is undertaken to ensure that QMS changes are effectively implemented. The requirements for change management is present in the following sections of ISO 9001:2015:
- 4.4 QMS and its processes;
- 5.3 Organizational roles, responsibilities and authorities;
- 7.5 Control of documented information;
- 8.1 Operational planning and control;
- 8.2.4 Changes to the requirements of products and services;
- 8.3.6 Design and development changes;
- 8.5.6 Control of changes.
You should seek and record evidence that your organization has retained documented information relating to planning and implementing changes that impact upon the management system.
Check that organizational knowledge is reviewed before changes to the QMS are made when responding to any change. Ensure that your organization has planned how to integrate and implement the changes into its QMS processes. Check that your organization has considered:
- The purpose of the changes and their potential consequences (risk and opportunities);
- The integrity of the management system (how does the change effect current process?);
- The availability of resources (are resources allocated to affect change?);
- The allocation or reallocation of responsibilities and authorities (are the right staff allocated?).
More information on PDCA
Planning
Context
Planning
Support
Doing
Support
Operations
Checking
Monitoring, measurement, analysis and evaluation
Acting
Improvement
Want to know more?
- Read our customer's feedback
- Client list - who's using our templates?
- How the templates are formatted and download examples
- Why we use turtle diagrams and process maps
- What's the difference between a process and a procedure?
- About documented information
SSL certification
A certificate guarantees the information your internet browser is receiving now originates from the expected domain - https://www.iso9001help.co.uk. It guarantees that when you make a purchase, sensitive data is encrypted and sent to the right place, and not to a malicious third-party.
Free PDCA guidance
ISO Navigator™ is our FREE online training tool that shows you how to apply the principles of PDCA to your operations. We also offer many helpful templates that get you on the road to documenting your management system, please visit the download page.
